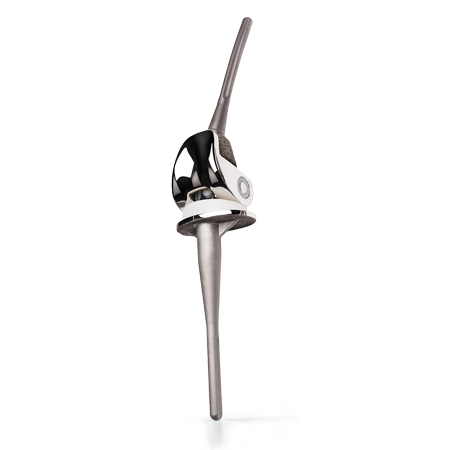
Limb Salvage
Zimmer Biomet’s Limb Salvage Portfolio was designed to address patients with severe bone loss, atypical joint deformities, multiple revisions or oncologic musculoskeletal disorders found in the hip, knee and shoulder. Our salvage systems are designed to provide surgeons with modularity and surgical latitude to best meet the unique needs of each individual patient.
OSS™ Orthopedic Salvage System
Zimmer Biomet’s Orthopedic Salvage System is designed to deliver a comprehensive solution for significant bone loss. Its modular platform offers surgeons intraoperative flexibility often required during challenging reconstructions.
Systems for Use with the OSS™ Orthopedic Salvage System
To increasingly meet the distinct needs of each patient, the Orthopedic Salvage System’s universal taper allows for complete system interchangeability between supplemental components providing more solutions to difficult revision cases.
Zimmer® Segmental System
The Zimmer® Segmental System is designed to address patients with severe bone loss associated with disease, trauma or revision. Cross-system capabilities with the NexGen® RH Knee provide the flexibility necessary for challenging reconstruction.
Tailored resources for your patients
Find videos, articles, and interactive content to guide your patients throughout their surgical journey on ReadyPatient.com, our dedicated patient recovery site.

All content herein is protected by copyright, trademarks and other intellectual property rights, as applicable, owned by or licensed to Zimmer Biomet or its affiliates unless otherwise indicated, and must not be redistributed, duplicated or disclosed, in whole or in part, without the express written consent of Zimmer Biomet.
This material is intended for health care professionals. Distribution to any other recipient is prohibited.
For product information, including indications, contraindications, warnings, precautions, potential adverse effects and patient counseling information, see the instructions for use or contact your local representative; search this website for additional product information. To obtain a copy of the current Instructions for Use (IFU) for full prescribing and risk information, please visit labeling.zimmerbiomet.com or call 1-800-348-2759, press 4 for 411 Technical Support.