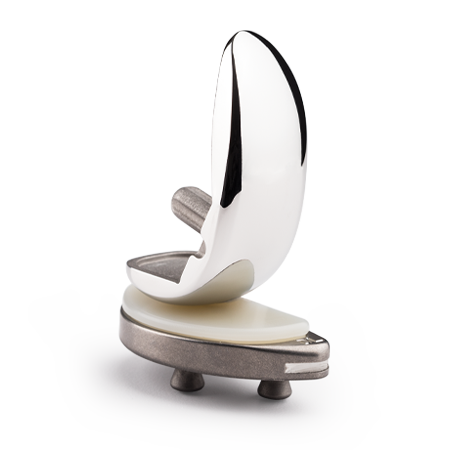
Persona® OsseoTi®Keel Tibia for Cementless Total Knee Arthroplasty
STABLE.1* VERSATILE. ANATOMIC.

The Persona OsseoTi Keel Tibia is the latest cementless tibia for total knee arthroplasty (TKA) within our clinically proven Persona Knee System. 2-3
STABLE
STABLE initial and biological fixation with OsseoTi porous keel design1*
VERSATILE
VERSATILE system design allows for selection between cementless and cemented procedures up until final implantation
ANATOMIC
ANATOMIC tibia for proper rotation and optimal bone coverage from the clinically proven Persona Knee System2-3
Product Features

- Spiked keel design provides stable initial mechanical fixation3 through the progressive press fit total knee implant design of the keel and pegs
- The anatomic tibia implant is designed so you no longer need to make compromise, facilitating both proper rotation and optimal bone coverage
- Switch between cementless or cemented procedures intraoperatively up until final implantation with a singular instrument tray
- OsseoTi Porous Metal Technology 3D printed structure directly mimics the architecture of cancellous bone, demonstrating excellent integration with host bone as early as 4 weeks in an animal study4**
- Medial Congruent (MC) bearing enhances medial conformity, improves knee stability, and the patient’s feeling of confidence while demonstrating higher patient satisfaction scores than the CR bearing5-6
Brochures
Videos
Hear Dr. Max Courtney's experience with Persona® OsseoTi® Keel Tibia
Related Products
SUBMIT YOUR INFORMATION AND GET CONTACTED BY A ZIMMER BIOMET REP
Legal Disclaimer
All content herein is protected by copyright, trademarks and other intellectual property rights, as applicable, owned by or licensed to Zimmer Biomet or its affiliates unless otherwise indicated, and must not be redistributed, duplicated or disclosed, in whole or in part, without the express written consent of Zimmer Biomet.
This material is intended for health care professionals. Distribution to any other recipient is prohibited.
For product information, including indications, contraindications, warnings, precautions, potential adverse effects and patient counseling information, see the instructions for use or contact your local representative; search this website for additional product information. To obtain a copy of the current Instructions for Use (IFU) for full prescribing and risk information, please visit labeling.zimmerbiomet.com or call 1-800-348-2759, press 4 for 411 Technical Support.