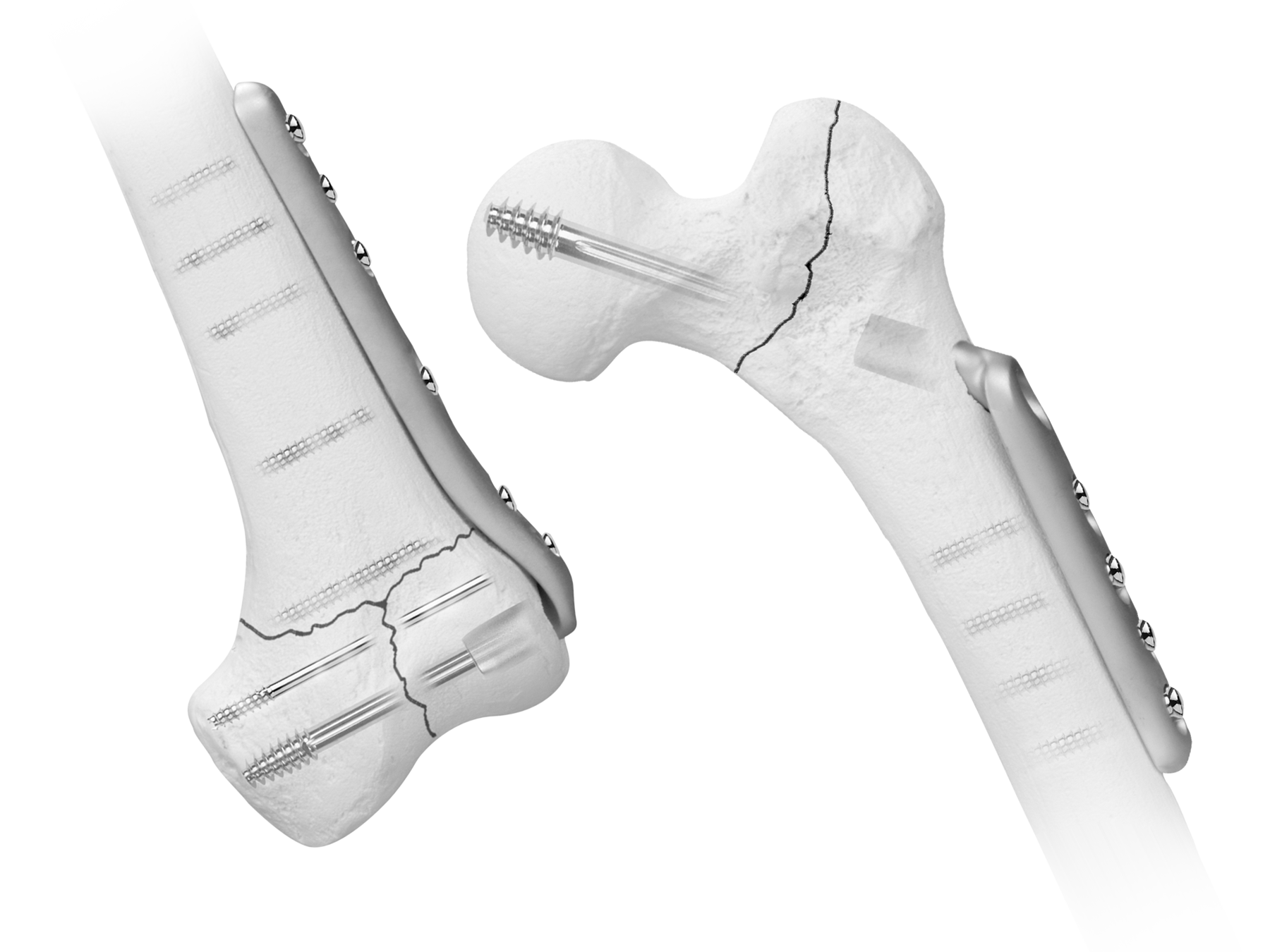
Versa-Fx® II Femoral Fixation
Combining strength with a tube/plate junction
The Versa-Fx® Femoral Fixation System may be used for the internal fixation of supracondylar fractures with displaced intra-articular fragments, with vertical intra-articular extension, and in the patient with multiple lower extremity fractures.
The Versa-Fx® II Femoral Fixation System combines the strength of 22-13-5 stainless steel with a tube/plate junction. Anatomically contoured, the plate is designed to provide optimum flexibility in angle fixation.
A wide selection of sizes, tube/plate angles, and screw hole configurations provide the versatility to use the Versa-Fx II System in a variety of indications, including intracapsular, intertrochanteric, and subtrochanteric fractures (Winquist Type Ill comminuted fractures). The tube plate is available in both keyed and keyless options, with a single set of instruments used for both.
Anatomy
- Hip & Femur
Product Type
- Plates and Screw
System Features
Benefits
Accurate
- Pin relocator provides accurate reinsertion of guide pin
Variable Options for Optimal Fit
- System offers a variety of tube angles that range between 130°-150° Compression Tube Plates and 95° or 90° Supracondylar Tube Plates with keyed and keyless tube plate design options. A choice of three thread options combined with a tapered core helps maximize the bone implant interface.
Tailored resources for your patients.
Find videos, articles, and interactive content to guide your patients throughout their surgical journey on ReadyPatient.com, our dedicated patient recovery site.
Legal Disclaimer
All content herein is protected by copyright, trademarks and other intellectual property rights, as applicable, owned by or licensed to Zimmer Biomet or its affiliates unless otherwise indicated, and must not be redistributed, duplicated or disclosed, in whole or in part, without the express written consent of Zimmer Biomet.
This material is intended for health care professionals. Distribution to any other recipient is prohibited.
For product information, including indications, contraindications, warnings, precautions, potential adverse effects and patient counseling information, see the instructions for use or contact your local representative; search this website for additional product information. To obtain a copy of the current Instructions for Use (IFU) for full prescribing and risk information, please visit labeling.zimmerbiomet.com or call 1-800-348-2759, press 4 for 411 Technical Support.