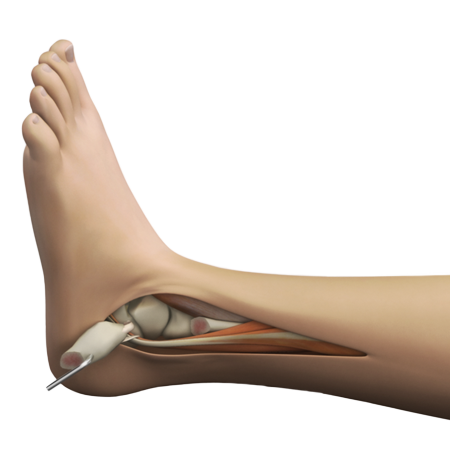
Lateral transfibular approach for peace of mind
Trabecular Metal™ Total Ankle
In Harmony with Natural Anatomy
Created to meet surgeon and patient needs, this ankle implant features a low profile design, lateral surgical approach and industry-first Prolong® Highly Crosslinked Polyethylene.
Anatomy
- Ankle
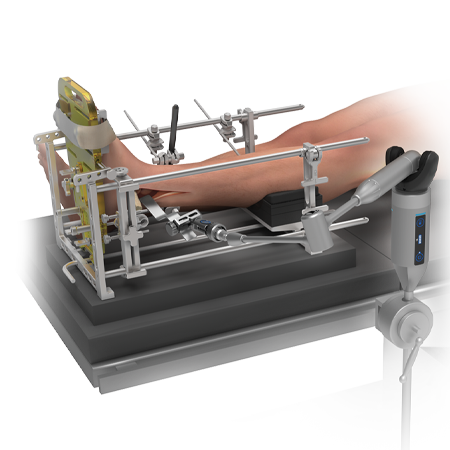
Procedure
- Total Ankle Replacement
Advanced implant designed to last
- Prolong Highly Crosslinked Polyethylene bearing surface to reduce volumetric wear.3,4
- Trabecular Metal has a high coefficient of friction vs. cancellous bone for stable initial fixation.5
- Reduced bony resection with the TM Ankle leaves more bone, which Statistically significantly increases bony support when compared to the alternative, flat resection.2


System Features
Specifications
Benefits
Durability
- The TM Ankle demonstrates 97.3% implant survivorship at three year follow-up, favorable to the overall class of TAA.1,6
Education
Literature
Videos
Trabecular Metal™ Total Ankle Animated Demonstration
Watch animation
Additional Information
Related Products
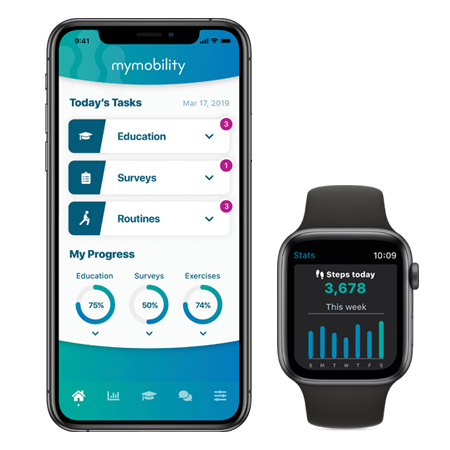
Tailored resources for your patients.
Find videos, articles, and interactive content to guide your patients throughout their surgical journey on ReadyPatient.com, our dedicated patient recovery site.
Legal Disclaimer
All content herein is protected by copyright, trademarks and other intellectual property rights, as applicable, owned by or licensed to Zimmer Biomet or its affiliates unless otherwise indicated, and must not be redistributed, duplicated or disclosed, in whole or in part, without the express written consent of Zimmer Biomet.
This material is intended for health care professionals. Distribution to any other recipient is prohibited.
For product information, including indications, contraindications, warnings, precautions, potential adverse effects and patient counseling information, see the instructions for use or contact your local representative; search this website for additional product information. To obtain a copy of the current Instructions for Use (IFU) for full prescribing and risk information, please visit labeling.zimmerbiomet.com or call 1-800-348-2759, press 4 for 411 Technical Support.