A convenient, modular system for post-traumatic elbow arthroplasty.
L2L® Radial Head System
Mobility Through Rotation
Designed with a smooth stem to be implanted line-to-line
Procedures
- Elbow Arthroplasty
Philosophies
- Ease of Insertion
- Cement-free
Application
- Severe Fracture
System Features
Specifications
Benefits
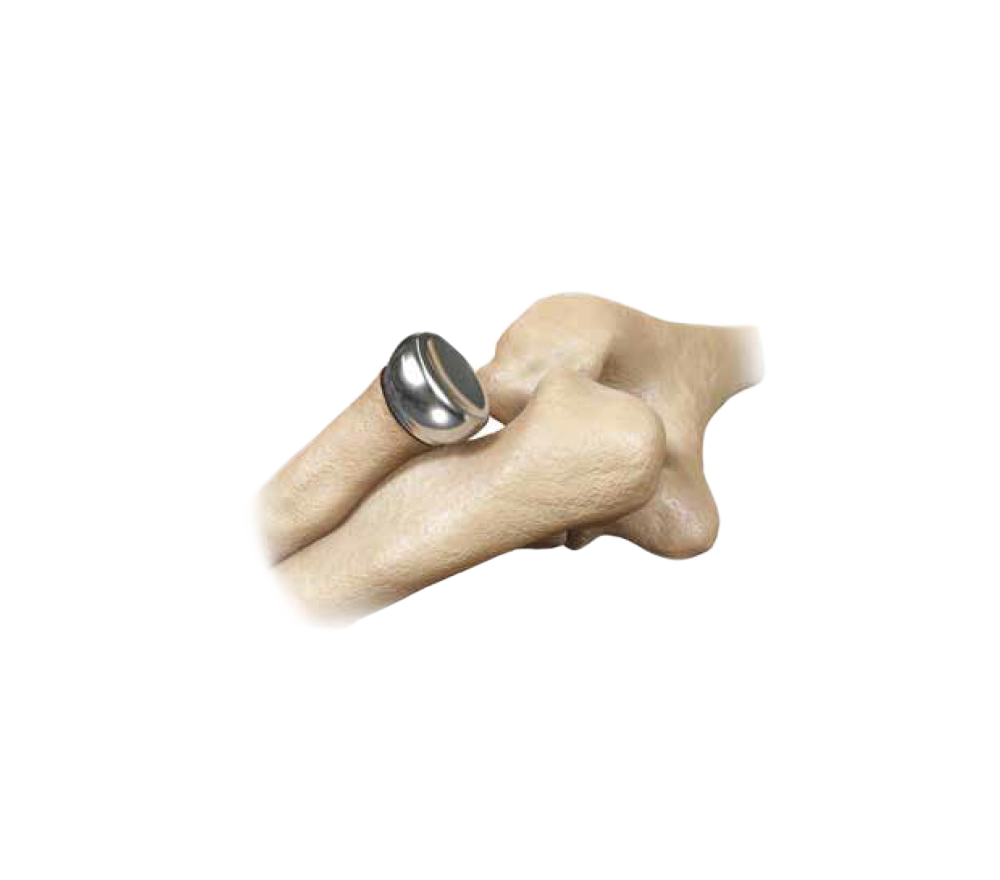
Smooth Motion
- The design of the radial head implant allows for smooth motion within the canal and facilitates easy movement in soft tissues during pronation and supination and is intended to restore stability in the joint.
Not cemented and Not press-fit
- Allows the Implant to act as a metal spacer with a design that incorporates sizing options to reproduce patient anatomy.1,2,3

Education
Literature
Additional Information
Related Products
Tailored resources for your patients.
Find videos, articles, and interactive content to guide your patients throughout their surgical journey on ReadyPatient.com, our dedicated patient recovery site.
All content herein is protected by copyright, trademarks and other intellectual property rights, as applicable, owned by or licensed to Zimmer Biomet or its affiliates unless otherwise indicated, and must not be redistributed, duplicated or disclosed, in whole or in part, without the express written consent of Zimmer Biomet.
This material is intended for health care professionals. Distribution to any other recipient is prohibited.
For product information, including indications, contraindications, warnings, precautions, potential adverse effects and patient counseling information, see the instructions for use or contact your local representative; search this website for additional product information. To obtain a copy of the current Instructions for Use (IFU) for full prescribing and risk information, please visit labeling.zimmerbiomet.com or call 1-800-348-2759, press 4 for 411 Technical Support.


