ZBX™ Zimmer Biomet
ASC Solutions

DELIVERING THE BUILDING BLOCKS FOR
OPERATIONAL EXCELLENCE
ZBX is Zimmer Biomet’s comprehensive ASC solutions program, offering you an experienced partner to strategically incorporate solutions, unique to your needs, providing efficiency in the OR and beyond.
EFFICIENT
Programs and services combined with world-class technology, designed to enhance operative flow, capacity and profitability.
EXPERIENCED
One partner to simplify the process from concept to completion, including financing and operational management solutions that grow with your practice.
STRATEGIC
Expertise and strategic partnerships to scale ASC programs, supporting operational and financial growth goals.

Real Estate Acquisition and Infrastructure Funding
The first step is understanding your Ambulatory Surgery Center (ASC) business plan and completing a business feasibility model through a proforma. If you don’t own land, we’ll introduce you to our strategic ambulatory surgery center real estate partner that can support you with the right capital opportunities and prepare a local market analysis to identify your ideal ASC location.
Design and Build
Already own the land? Through our partnerships, choose from ambulatory surgery center services such as developer consultation and/or full-service project management to oversee construction from foundation to final touches.

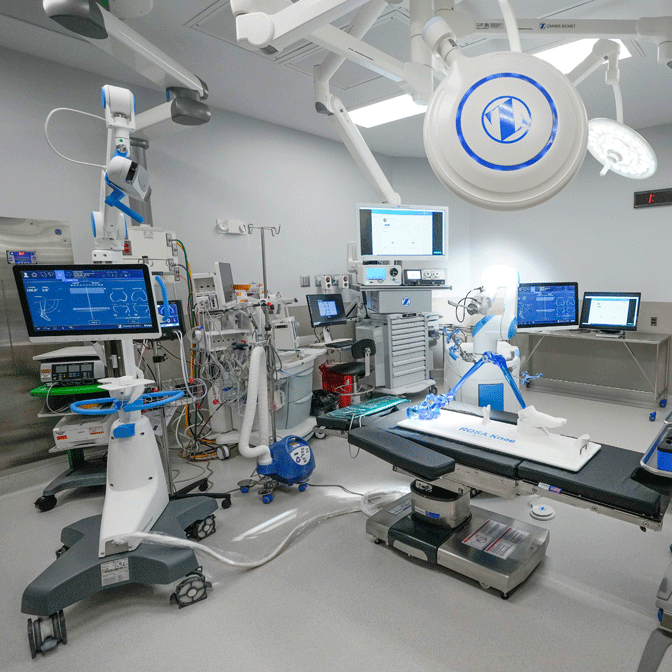
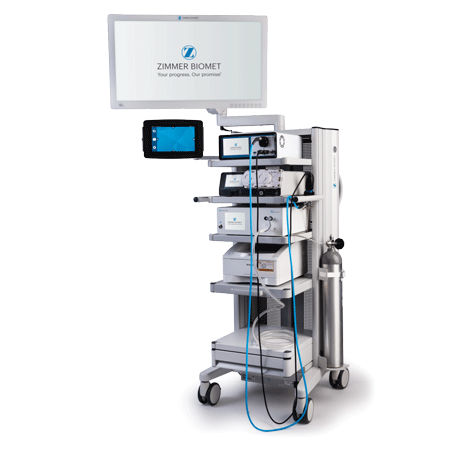
Capital Equipment
Outfit your OR with ASC equipment needed to enhance surgical outcomes and optimize your surgery workflow that supports your care teams and helps deliver exceptional patient care. Now offering lights, booms, beds, SPD, and infection control, all available with flexible financing solutions tailored to you.
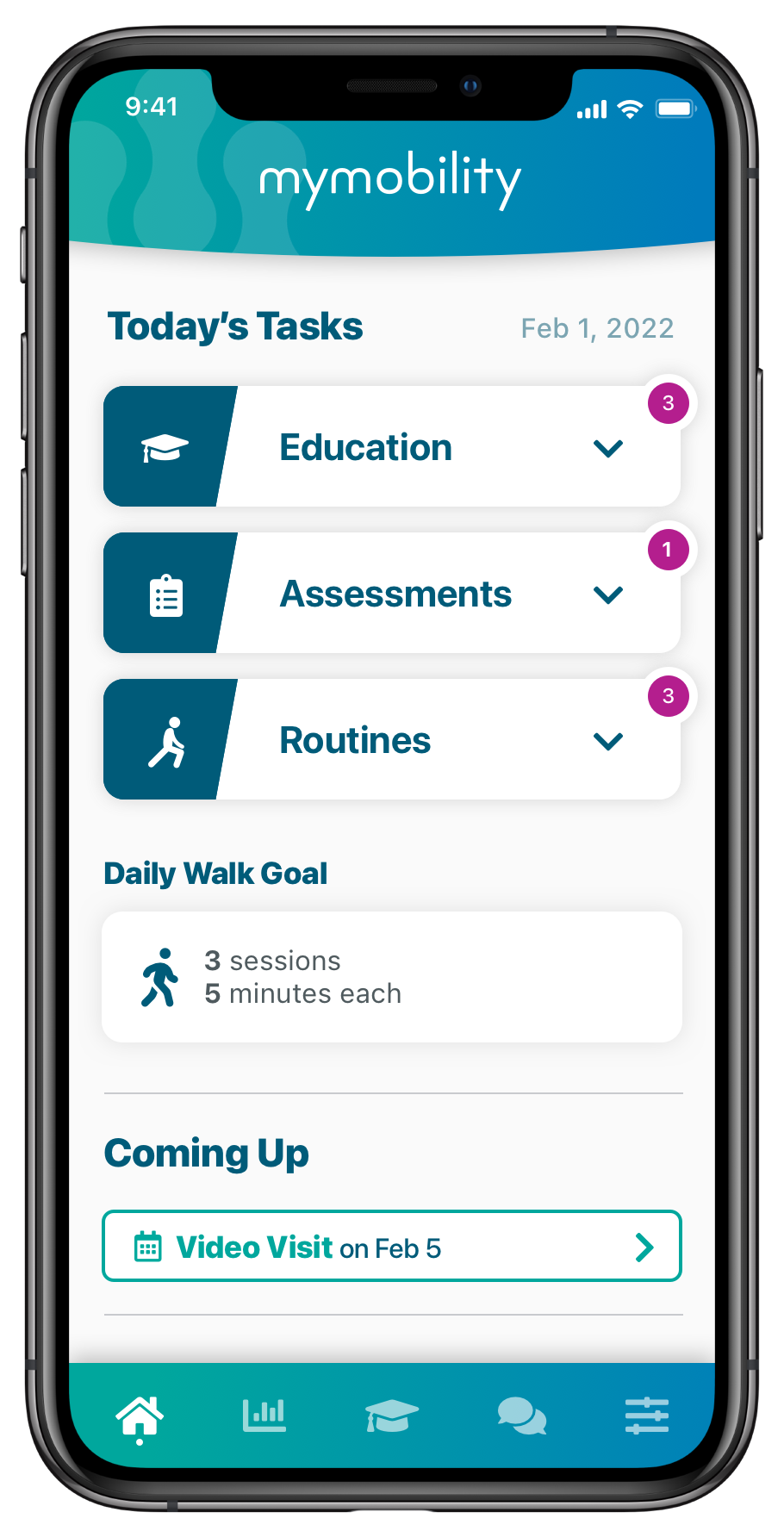
Operational Efficiency
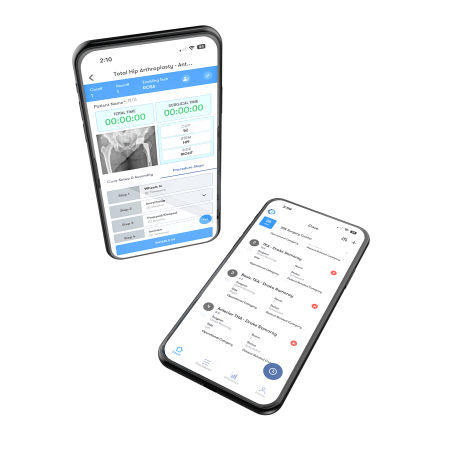
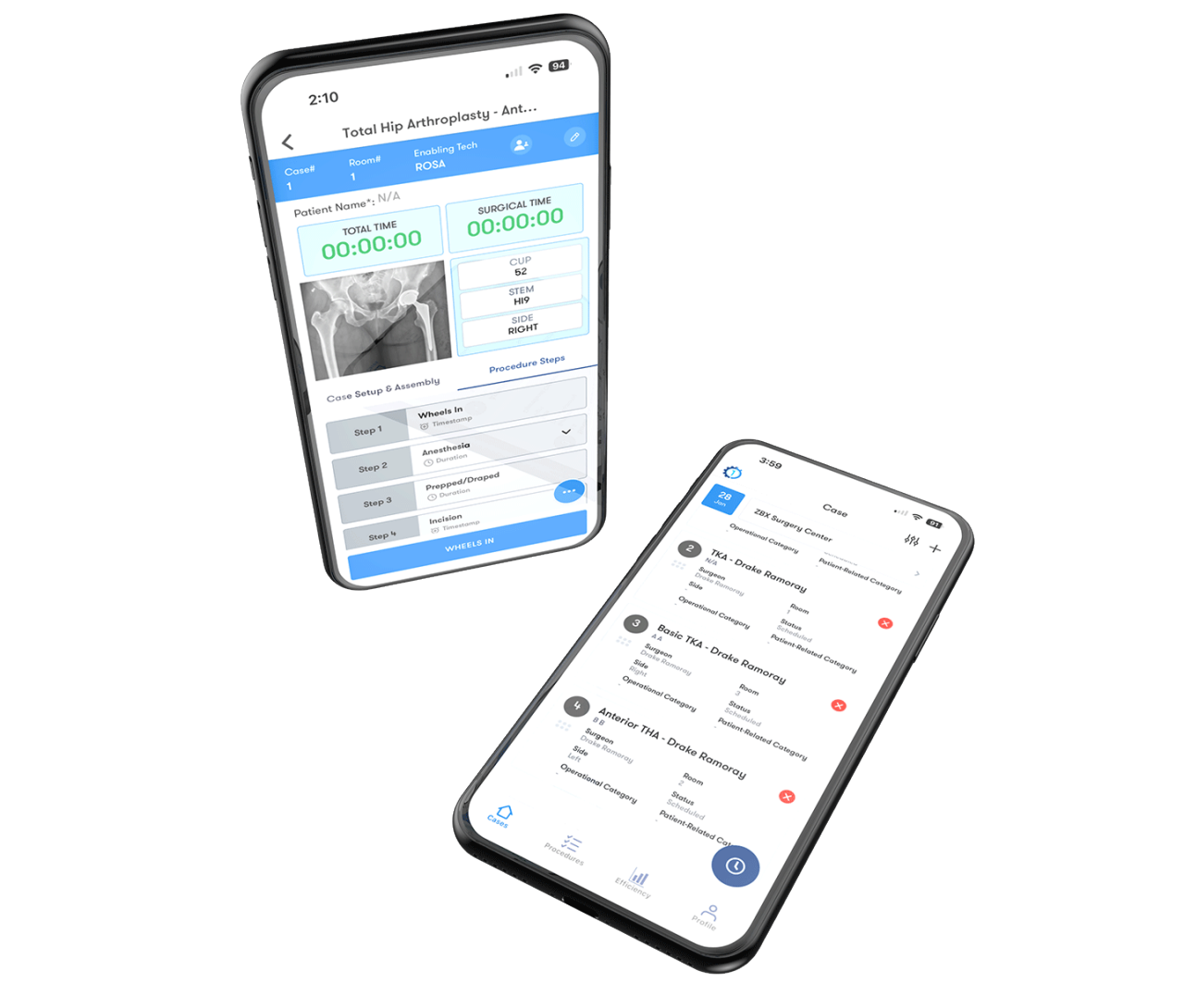
1Team® OR Efficiency Software equips surgeons and their care teams with procedure-specific metrics designed to enhance peri-operative efficiency and offers customizable reports to drive maximum change.
For a demo of 1Team, our healthcare software solution, schedule here.
Related Products
NOT READY TO BOOK AN APPOINTMENT, BUT WANT MORE INFORMATION ON ZBX?
COMPLETE THE FORM TO LEARN MORE
Disclaimer
This material is intended for healthcare professionals. Distribution to any other recipient is prohibited. Zimmer Biomet does not practice medicine. For indications, contraindications, warnings, precautions, potential adverse effects and patient counselling information, see the Instructions for Use or contact your local representative; visit www.zimmerbiomet.com for additional product information.