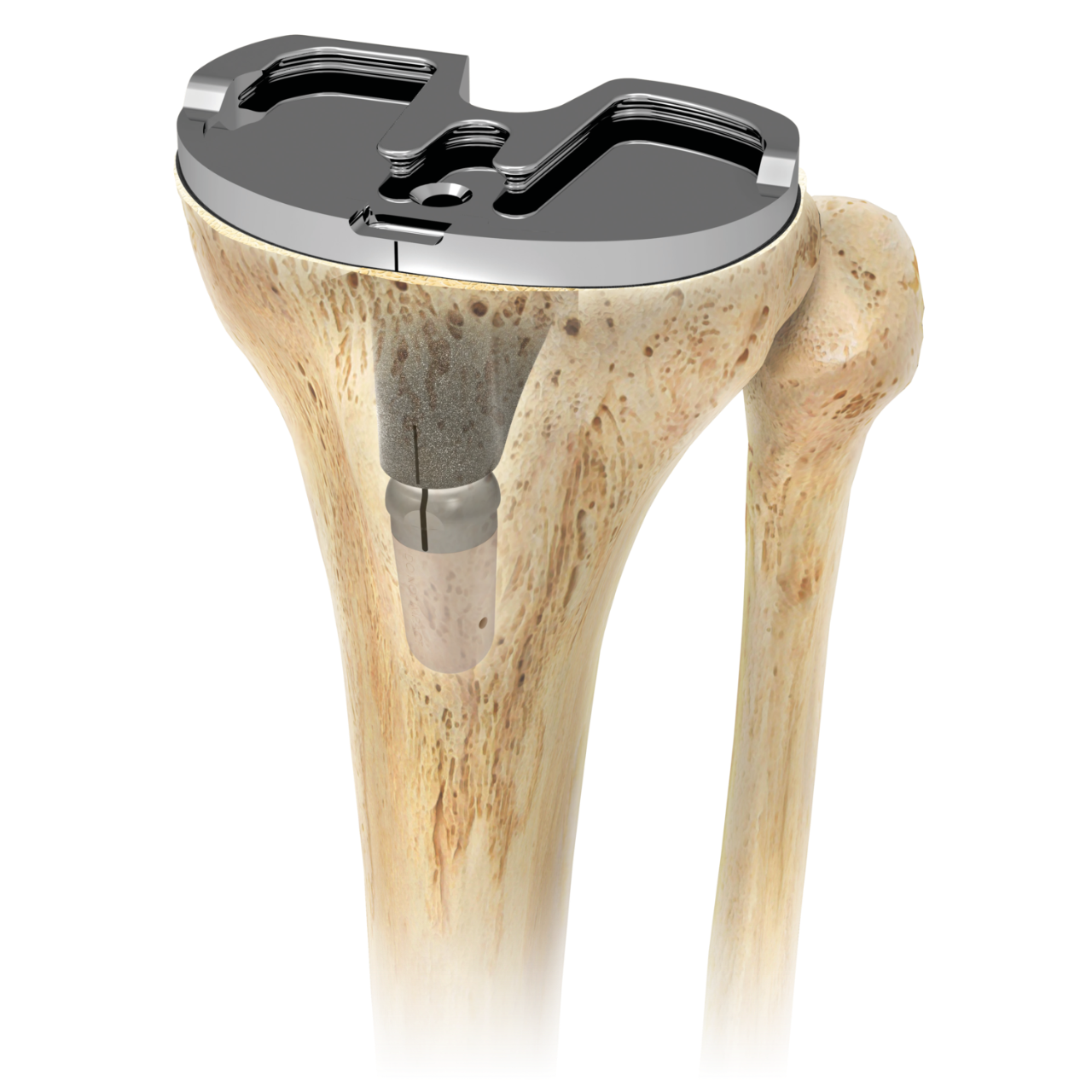
Persona IQ®
The Smart Knee®

Persona IQ® The Smart Knee® , now available in a 30 mm stem, collects data directly from the knee to provide post-operative recovery insights1,2 through the mymobility® Care Management platform, allowing care teams to monitor and personalize the TKA patient experience1,2,3 with the goal of improving practice efficiency4-7 .
Persona IQ Customer Testimonials
See what patients are saying about Persona IQ
Persona IQ Brand Anthem Video
Discover how we are taking the next steps in orthopedic innovation.
Automatically collects and transmits gait and range of motion metrics to the mymobility® Care Management Platform, to provide insights into patient recovery trends.
Patients experience a connected and personalized recovery knowing their care team has remote access to view their specific recovery data, 24/7.
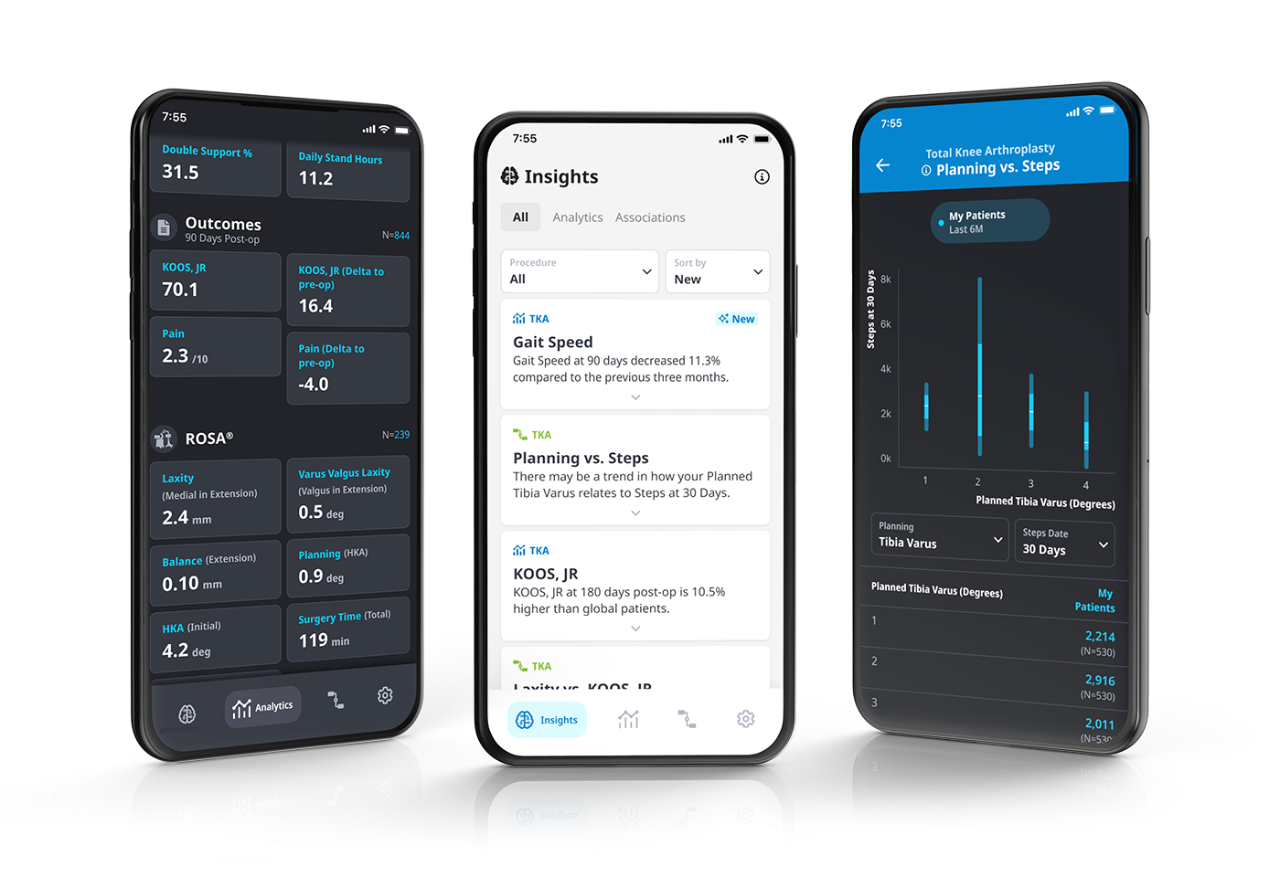
Through intuitive dashboards, easily monitor patient data based on recovery thresholds, providing an efficient way to analyze and review recovery trends.
Data Collection
Persona IQ is a first-to-world smart knee implant that captures patient-specific gait and range of motion metrics during the course of patient monitoring.
Secure Data Flow
The collection and utilization of patient-specific data is easy with Persona IQ.
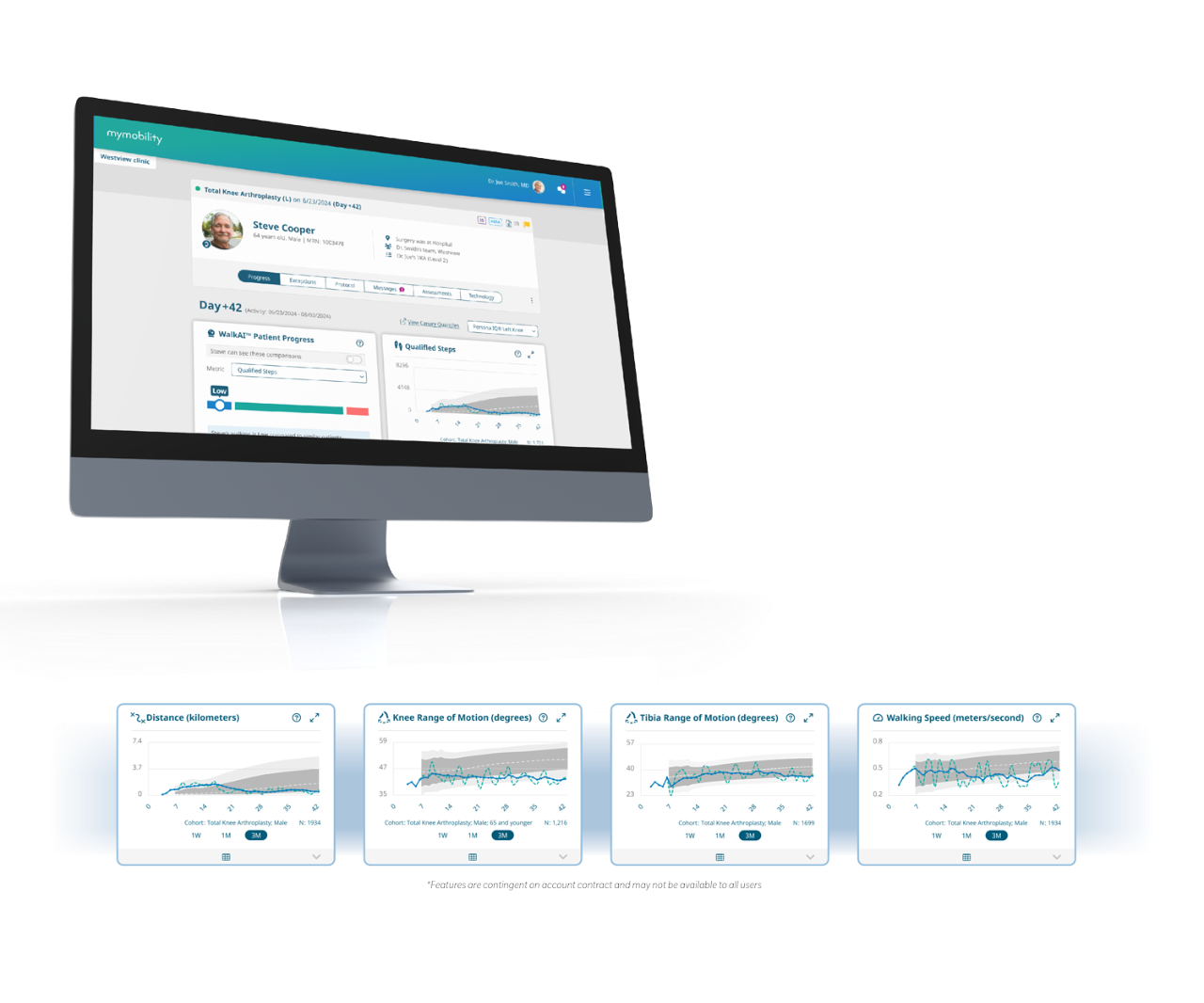
The post-operative data is available through the mymobility® Care Management Platform, offering both patients and physicians a simple to use intuitive interface.

How it Works
Monitor and personalize the Persona IQ patient experience with the mymobility® Care Management Platform
mymobility provides support and guidance to patients through a connected experience collecting the following metrics. All you have to do is enroll.
- Education & Engagement
- PROMs Collection Exceptions (Pain & Gait)
- Effortless Activity + Physiologic Tracking
- Pre- and Post-Operative Exercises
- Telemedicine (Virtual Visits) & Messaging
- Recovery Curve Data Analysis
- Patient Progress

Easily compare Persona IQ post-operative metrics to trend data from other Persona IQ patients.
Indications and Contraindications
Indications
The Canary Tibial Extension (CTE) with
Canary Health Implanted Reporting Processor (CHIRP) System is intended
to provide objective kinematic data from the implanted medical device
during a patient’s total knee arthroplasty (TKA) post-surgical care.
The kinematic data are an adjunct to other physiological parameter
measurement tools applied or utilized by the physician during the
course of patient monitoring and treatment post-surgery.
The device is indicated for use in patients undergoing a cemented TKA procedure that are normally indicated for at least a 58 mm sized tibial stem extension.
The objective kinematic data generated by the CTE with CHIRP System are not intended to support clinical decision-making and have not been shown to provide any clinical benefit. The CTE with CHIRP System is compatible with Zimmer Persona® Personalized Knee System.
Contraindications
The canturio™te (CTE) is contraindicated for use in patients who are
undergoing procedures or treatments at or in the proximity of the CTE
using therapeutic ionizing radiation can result in shortened battery
life or premature failure of electronic components. Damage to the CTE
by therapeutic ionizing radiation may not be immediately
detectable.
Before performing TKA on any patient, consider the following contraindications.
The Zimmer Biomet Persona® Knee System is contraindicated for use in patients who have—
- Previous history of infection in the affected joint and/or other local/systemic infection that may affect the prosthetic joint
- Insufficient bone stock on femoral or tibial surfaces
- Skeletal immaturity
- Neuropathic arthropathy
- Osteoporosis or any loss of musculature or neuromuscular disease that compromises the affected limb
- A stable, painless arthrodesis in a satisfactory functional position
- Severe instability secondary to the absence of collateral ligament integrity
Total Knee Arthroplasty is contraindicated in patients who have rheumatoid arthritis (RA) accompanied by an ulcer of the skin or a history of recurrent breakdown of the skin because their risk of postoperative infection is greater. RA patients using steroids may also have increased risk of infection. Late infections in RA patients have been reported 24+ months postoperative.
Privacy
We understand the importance of privacy and are dedicated to protecting that right. We only use customer and patient data to provide the services we’ve agreed upon and for purposes that will enable those services. Persona IQ is not designed to track patient location, and Zimmer Biomet will protect the data you entrust us with through strong safeguards.
Privacy
Security
Usage
Ownership
Additional Information
Have questions? Contact Smart Knee Support at SmartKneeSupport@zimmerbiomet.com or 1-844-799-8208”
Tailored resources for your patients
Find videos, articles, and interactive content to guide your patients throughout their surgical journey on ReadyPatient.com, our dedicated patient recovery site.
Videos
OR Base Station Demo
Learn more about the OR Base Station
Brochures
Reimbursement Information
Click here to view the Zimmer Biomet Coding
Guide for Persona IQ.
CONTACT US TO DISCOVER MORE ABOUT PERSONA IQ!
SUBMIT YOUR INFORMATION AND GET CONTACTED BY A ZIMMER BIOMET REP
Disclaimer
All content herein is protected by copyright, trademarks and other
intellectual property rights, as applicable, owned by or licensed to
Zimmer Biomet or its affiliates unless otherwise indicated, and must
not be redistributed, duplicated or disclosed, in whole or in part,
without the express written consent of Zimmer Biomet.
For indications, contraindications, warnings, precautions, potential adverse effects and patient counselling information, see the package insert or contact your local representative; visit www.zimmerbiomet.com for additional product information.
This material is intended for healthcare professionals. Distribution to any other recipient is prohibited.
Canary Medical, Canary Quantiles™, CANARY canturio™te, Canary Bird Logo and CHIRP™ are the exclusive property of Canary Medical or its affiliates.
For full prescribing information for the CANARY canturio ™ te, refer to physician instructions for use.
The Canary Quantiles™ Recovery Curves software provides health care professionals (HCPs) with additional aggregate population data when managing a patient’s total knee arthroplasty (TKA) post-surgical care. HCPs can filter or select options for additional views based on patient demographics (e.g. age), to analyze trends and outcomes. The Canary Quantiles Recovery Curves software allows HCPs to view aggregate patient population data to analyze patient recovery progress and direction of outcome. The Canary Quantiles Recovery Curves software does not control the function or parameters of the Canturio™ Tibial Extension (CTE) with Canary Health Implanted Reporting Processor (CHIRP®) System and is not intended for active patient monitoring. The information in this document is provided solely for the purpose of acquainting you with Canary Medical Inc. and its subsidiaries (the “Company”, “we”, “us” or “our”). This document does not constitute an offer or solicitation to sell any software or services, all of such services shall be subject to separate Terms of Use and/or subscription documentation. All trademarks are the exclusive property of their respective owners. Dr. Canturio is an imaginary healthcare provider with a fabricated surname.
All rights reserved. The information contained in this document is provided solely for the purpose of acquainting you, as its recipient, with Canary Medical and its subsidiaries (the “Company”, “we”, “us” or “our”) and its executive personnel. This document does not constitute an offer to sell, or a solicitation of an offer to buy, any securities of the Company, nor will we make any offer, solicitation or sale of such securities in any state, province or jurisdiction in which such an offer, solicitation or sale would be unlawful prior to registration or qualification under the securities laws of any such state or jurisdiction. All trademarks are the exclusive property of their respective owners. Dr. Canturio is an imaginary health care provided with a fabricated surname.
Patients must have compatible internet access and a text-capable mobile device, or a smartphone to use mymobility; not all smartphone app features are available with web-based version.
Not all patients are candidates for the use of this product and surgeons should evaluate individually to determine which patients are appropriate for therapy at home.
All names used in the mymobility app examples are fictitious. No
identification with actual patients or health care professionals is
intended or should be inferred. Apple, iPhone and Mac are registered
trademarks of Apple, Inc.