genex® Bone Graft Substitute is a simple to use synthetic absorbable material designed to promote regeneration of bone in osseous defects.
genex® Bone Graft Substitute
Complements the body's natural healing processes and encourages normal bone structure
Unrivaled flexibility for delivery and application
Completely absorbs to leave no trace
In 12 months, genex is completely absorbed and remodeled while leaving no foreign artifacts after resorption1,2
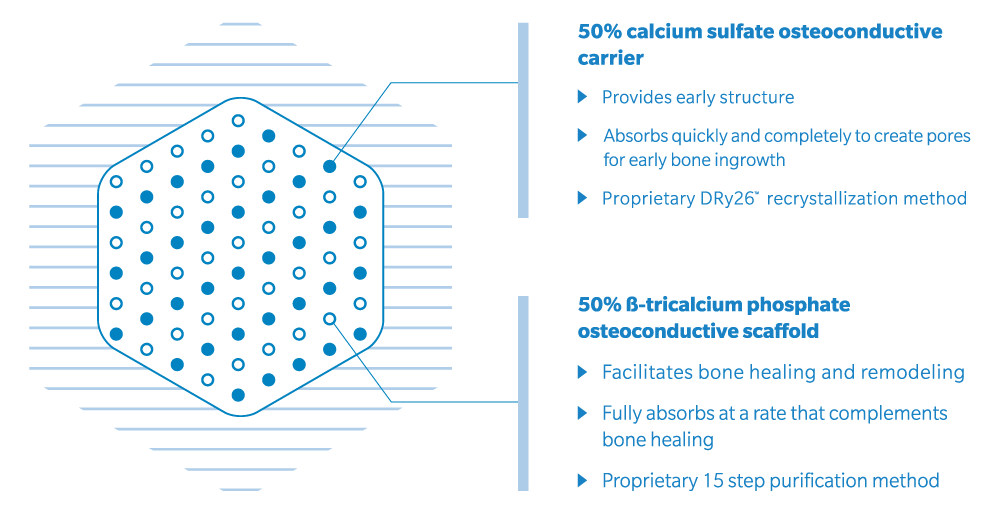
Biphasic composite of purity, balance, and characteristics
Our proprietary recrystallization and purification methods removes impurities from genex such as6
- No inflammatory pyrophosphates
- No slow and non-absorbing compounds such as hydroxyapatite
Several studies have concluded healthy bone is restored in a clinically relevant timeframe7-9
genex is a precisely balanced ß tricalcium phosphate/calcium sulfate hemihydrate compound with distinct design properties:
- Contains no hydroxyapatite (HA)
- Negatively charged surface chemistry
- Compressive strength similar to trabecular bone
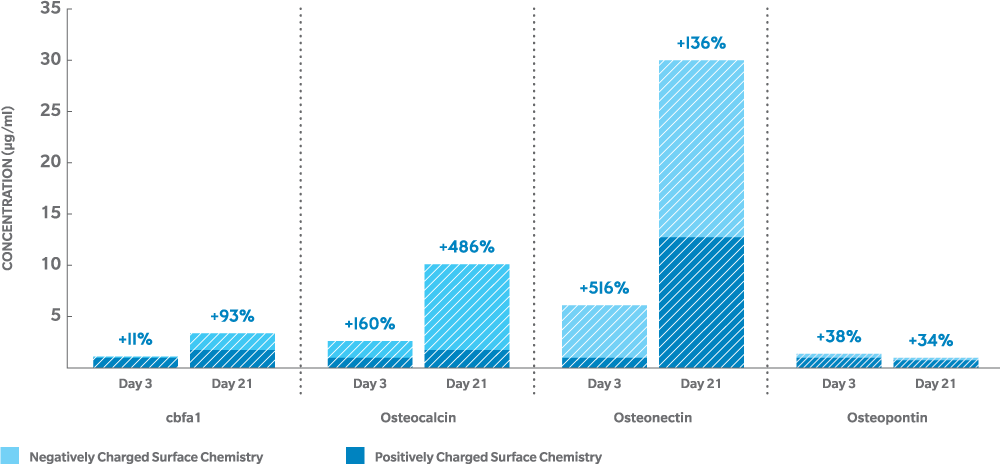
Negatively charged genex enhances the osteogenic response to accelerate bone growth10,14
- genex containts no hydroxyapatite.
- HA can only be absorbed at 1-2% per year12
- up to 5 times normal levels (on the image):
- In-vitro human osteoblast culture, 3 days11
System Features
- Unrivaled flexibility of delivery and application
- Closed-mixing system is 2x faster to prepare than open mixing system13
- Longer working time
- Drillable 15 minutes after mixing

Videos
genex® Trailer
Additional Information
Tailored resources for your patients.
Find videos, articles, and interactive content to guide your patients throughout their surgical journey on ReadyPatient.com, our dedicated patient recovery site.
Legal Disclaimer
All content herein is protected by copyright, trademarks and other intellectual property rights, as applicable, owned by or licensed to Zimmer Biomet or its affiliates unless otherwise indicated, and must not be redistributed, duplicated or disclosed, in whole or in part, without the express written consent of Zimmer Biomet.
This material is intended for health care professionals. Distribution to any other recipient is prohibited.
For product information, including indications, contraindications, warnings, precautions, potential adverse effects and patient counseling information, see the instructions for use or contact your local representative; search this website for additional product information. To obtain a copy of the current Instructions for Use (IFU) for full prescribing and risk information, please visit labeling.zimmerbiomet.com or call 1-800-348-2759, press 4 for 411 Technical Support.

