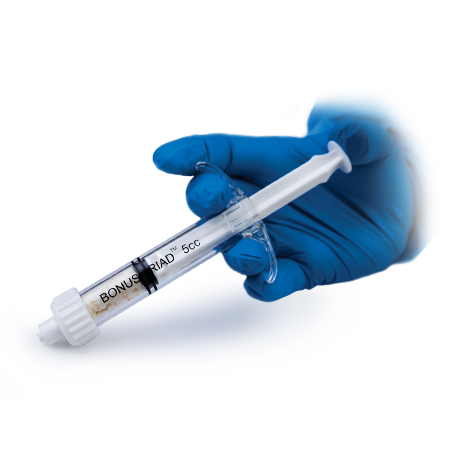
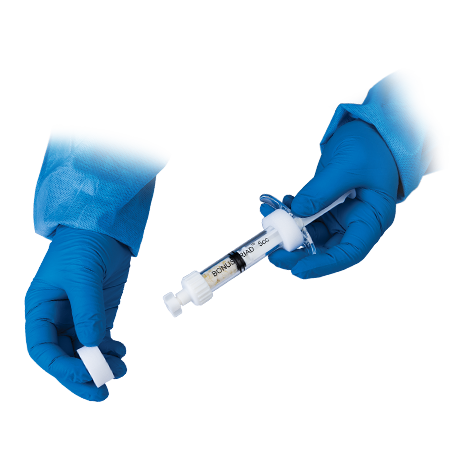
Complete bone remodeling
Bonus Triad™
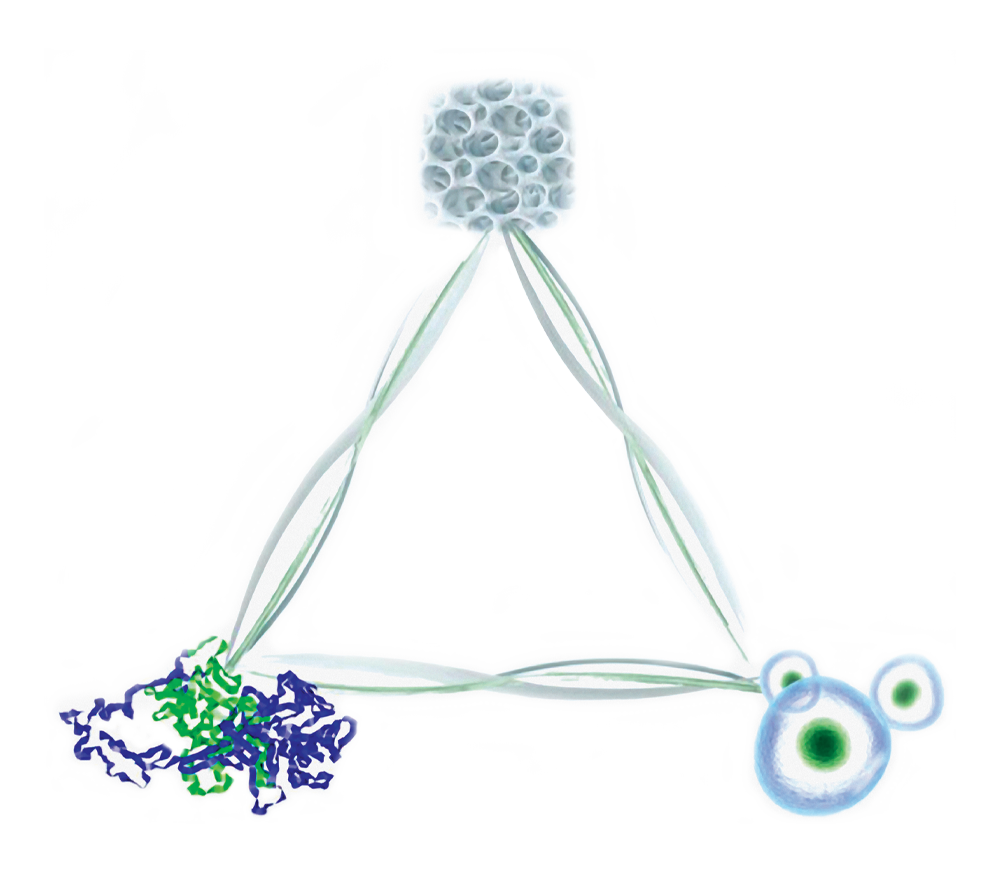
Elements of Bone Remodeling
Osteoconductive. Osteoinductive. Osteogenic.
Advanced fresh-frozen allografts were developed from cadaveric tissue to meet the need for an alternative to autograft. These allografts retain osteoconductive and osteoinductive properties as well as naturally adherent viable cells.1"
Anatomy
- Biologics
Product Type
- Mesenchymal Stem Cells
Bonus Triad
Every donor lot of Bonus Triad Allograft is tested to confirm that it contains
750,000 cells/ccof cancellous tissue with at least 70% cell viability.”


Product Features
Specifications
Benefits
Ease of Use
- Bonus Triad Allograft is packaged in an intuitive, proprietary pre-filled delivery syringe and features a built-in filter that allows for the full preparation of the material directly inside the syringe.
- Cryoprotective solution frozen at -70°C to preserve cell viability
Precise Handling
- The cortical fibers are demineralized and create a cohesive graft that stays together.4
Videos
Bonus Triad Intraoperative Use
Bonus Triad™ Animation”
Additional Information
Tailored resources for your patients.
Find videos, articles, and interactive content to guide your patients throughout their surgical journey on ReadyPatient.com, our dedicated patient recovery site.
Legal Disclaimer
All content herein is protected by copyright, trademarks and other intellectual property rights, as applicable, owned by or licensed to Zimmer Biomet or its affiliates unless otherwise indicated, and must not be redistributed, duplicated or disclosed, in whole or in part, without the express written consent of Zimmer Biomet.
This material is intended for health care professionals. Distribution to any other recipient is prohibited.
For product information, including indications, contraindications, warnings, precautions, potential adverse effects and patient counseling information, see the instructions for use or contact your local representative; search this website for additional product information. To obtain a copy of the current Instructions for Use (IFU) for full prescribing and risk information, please visit labeling.zimmerbiomet.com or call 1-800-348-2759, press 4 for 411 Technical Support.