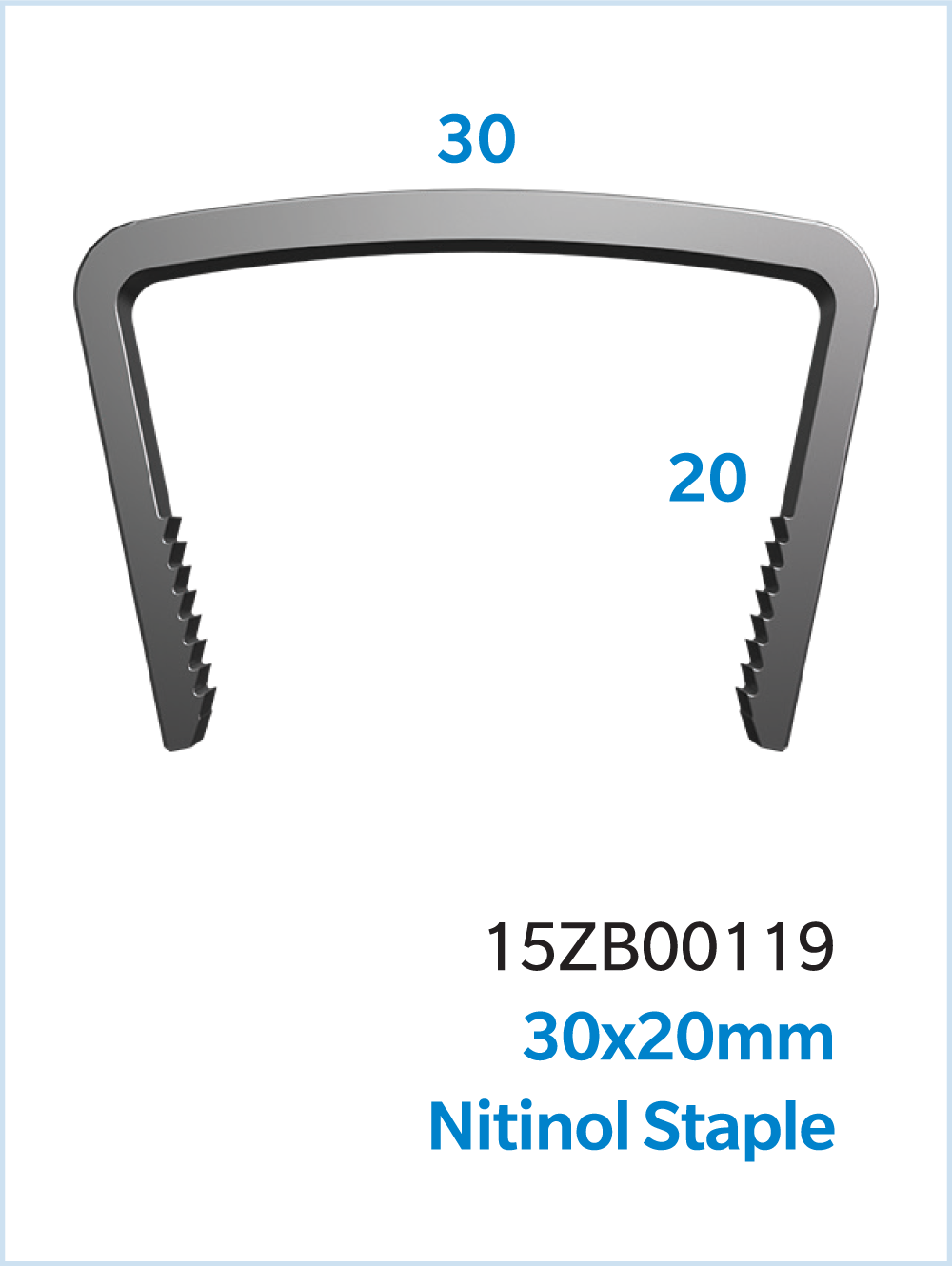
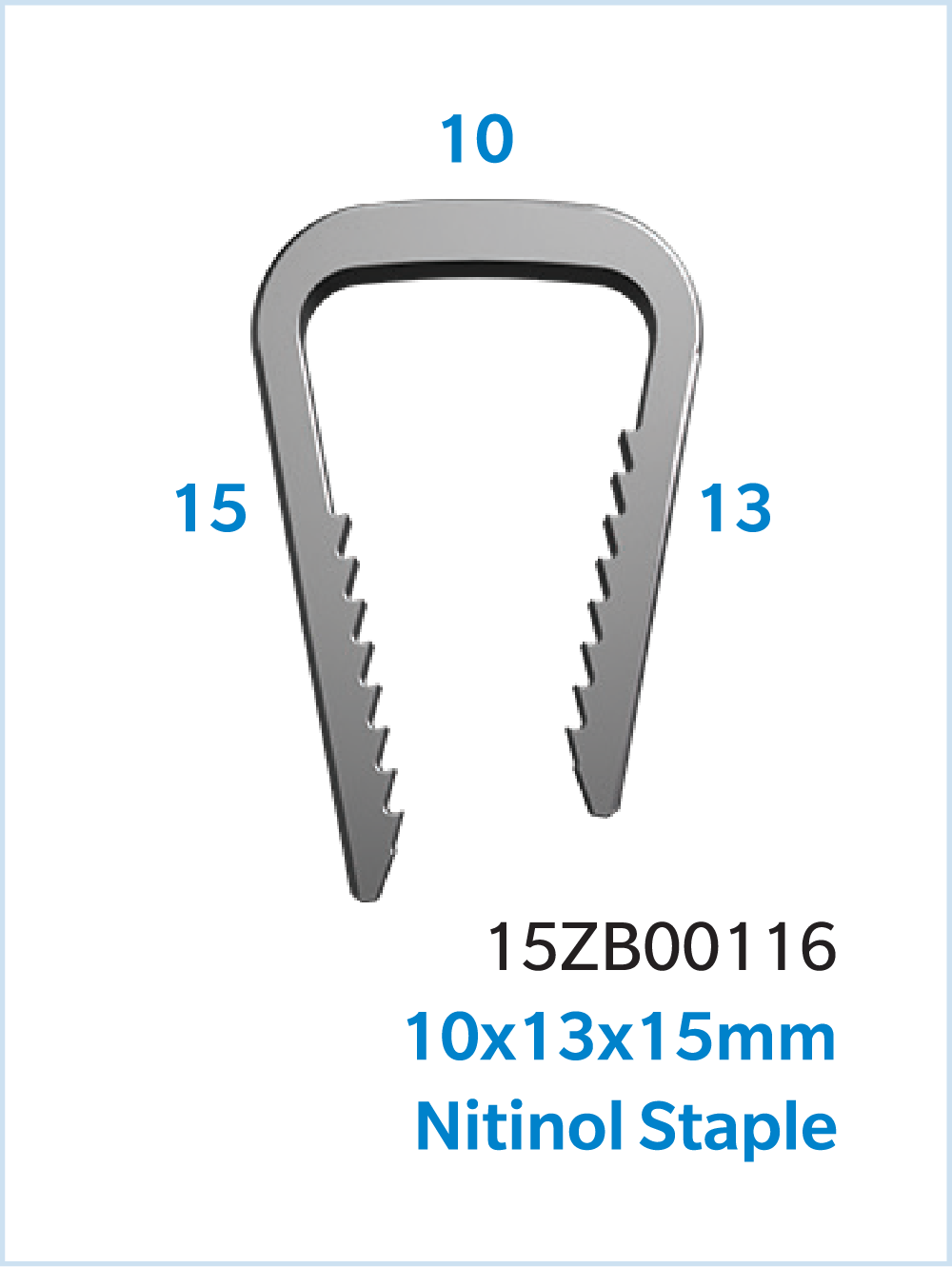
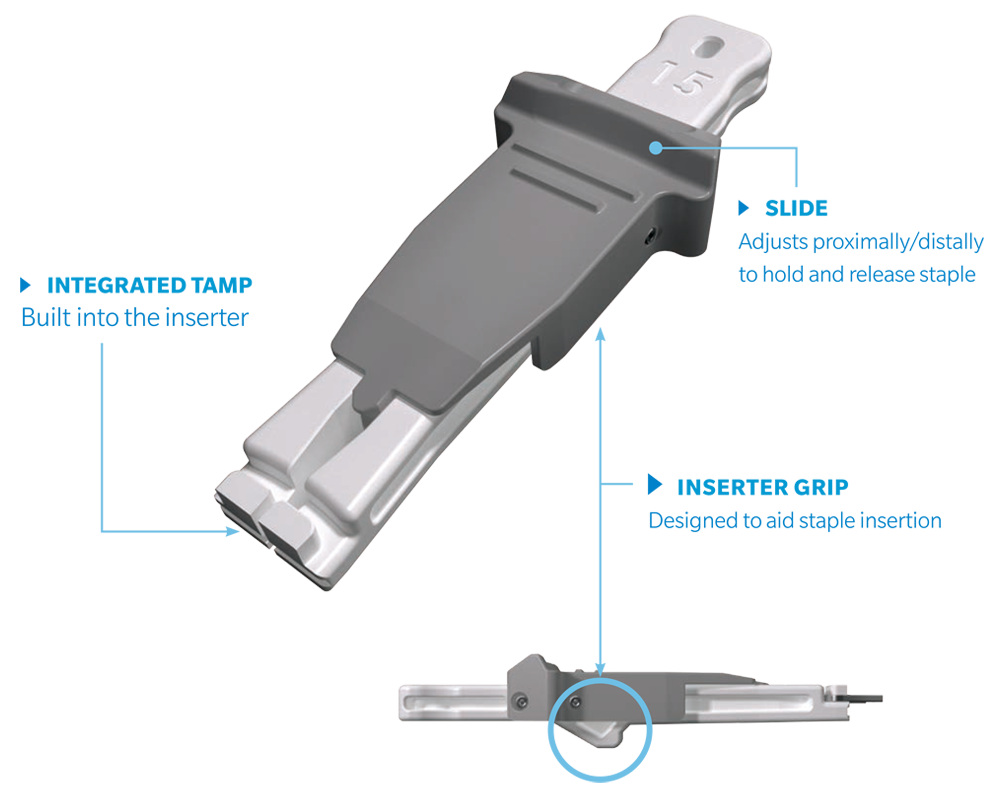
The UNITUS™ Staple System is a Nitinol compression staple with a self-contained, sterile instrument kit, and inserter grip designed to aid staple insertion. UNITUS Staples are available in multiple combinations of bridge width, leg lengths, and cross sections to accommodate various anatomies.
UNITUS™ Staple System
Overview
Designed for Efficiency
- Inserter designed with built in tamp
- Surgeon’s eyes do not have to leave field of view
- Inserter can aid intraoperative staple removal
Comprehensive Sizes for a variety of surgical needs
- Staple sizes ranging from 8mm x 8mm to 30mm x 20mm, including offset leg length staples
- Indications for:
- Hand and foot arthrodesis
- Proximal tibial metaphysis osteotomy
- Fixation of small bone fragments
Strength
- Fatigue Strength measured 4x more cycles at a 50% higher load than a leading competitive staple1
- Pull out strength measured 1.5x stonger than a leading competitive staple1
- Nitinol material designed to provide consistent compressive force across fixation site
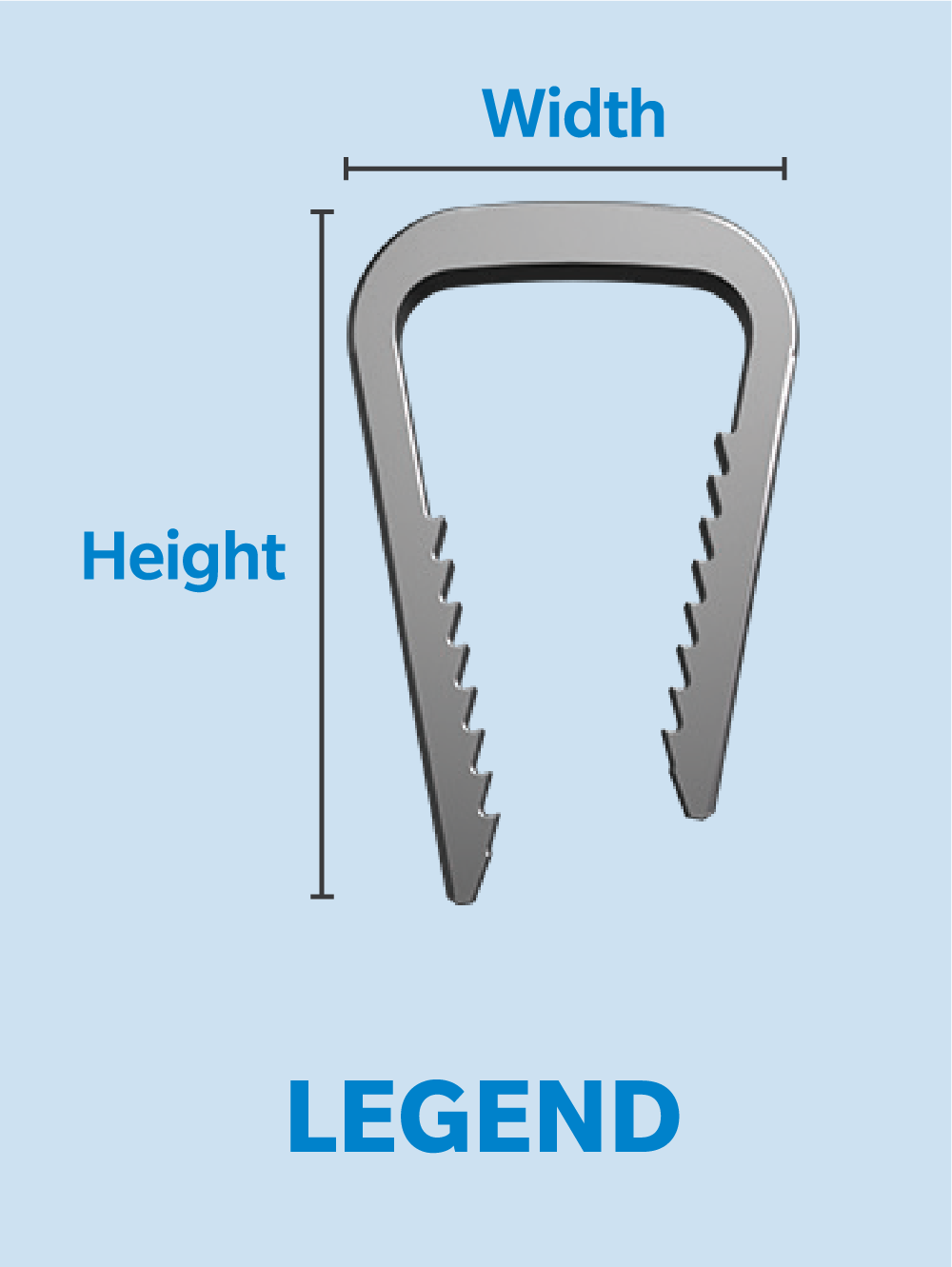
Specifications
- Multiple bridge widths and leg lengths.
- Single use inserter, drill guide, drill bit, and locating pins all included in each kit
Videos
UNITUS Technique Animation
Watch animation
Additional Information
Tailored resources for your patients.
Find videos, articles, and interactive content to guide your patients throughout their surgical journey on ReadyPatient.com, our dedicated patient recovery site.
Legal Disclaimer
All content herein is protected by copyright, trademarks and other intellectual property rights, as applicable, owned by or licensed to Zimmer Biomet or its affiliates unless otherwise indicated, and must not be redistributed, duplicated or disclosed, in whole or in part, without the express written consent of Zimmer Biomet.
This material is intended for health care professionals. Distribution to any other recipient is prohibited.
For product information, including indications, contraindications, warnings, precautions, potential adverse effects and patient counseling information, see the instructions for use or contact your local representative; search this website for additional product information. To obtain a copy of the current Instructions for Use (IFU) for full prescribing and risk information, please visit labeling.zimmerbiomet.com or call 1-800-348-2759, press 4 for 411 Technical Support.