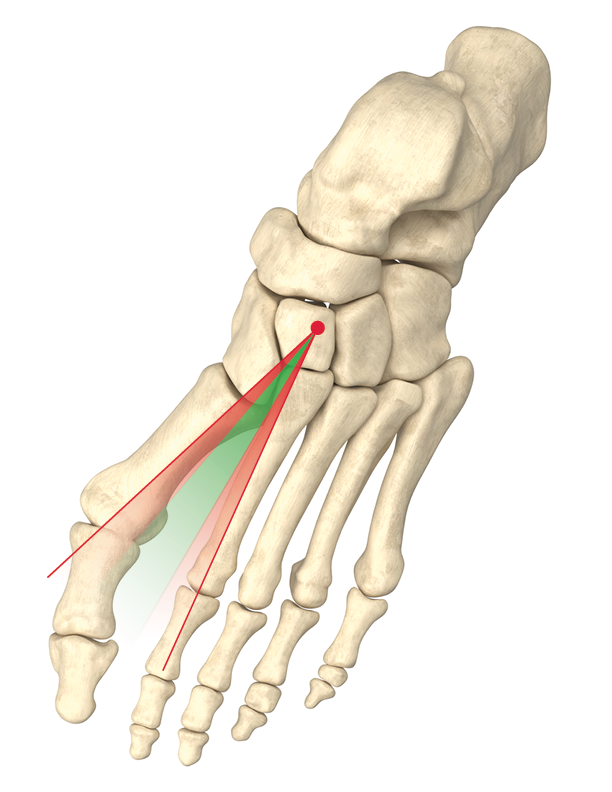
Provides corrections of intermetatarsal (IM) Angle and Frontal Plane Deformity, offering a seamless transition from templating to plating to produce precise, reproducible outcomes.
Axi+Line® Proximal Bunion Correction System
Correction
Provides corrections of intermetatarsal (IM) Angle and Frontal Plane Deformity
In one study of 166 feet, 87.3% of patients with hallux valgus had abnormal frontal plane rotation, also known as pronation, of the 1st metatarsal1
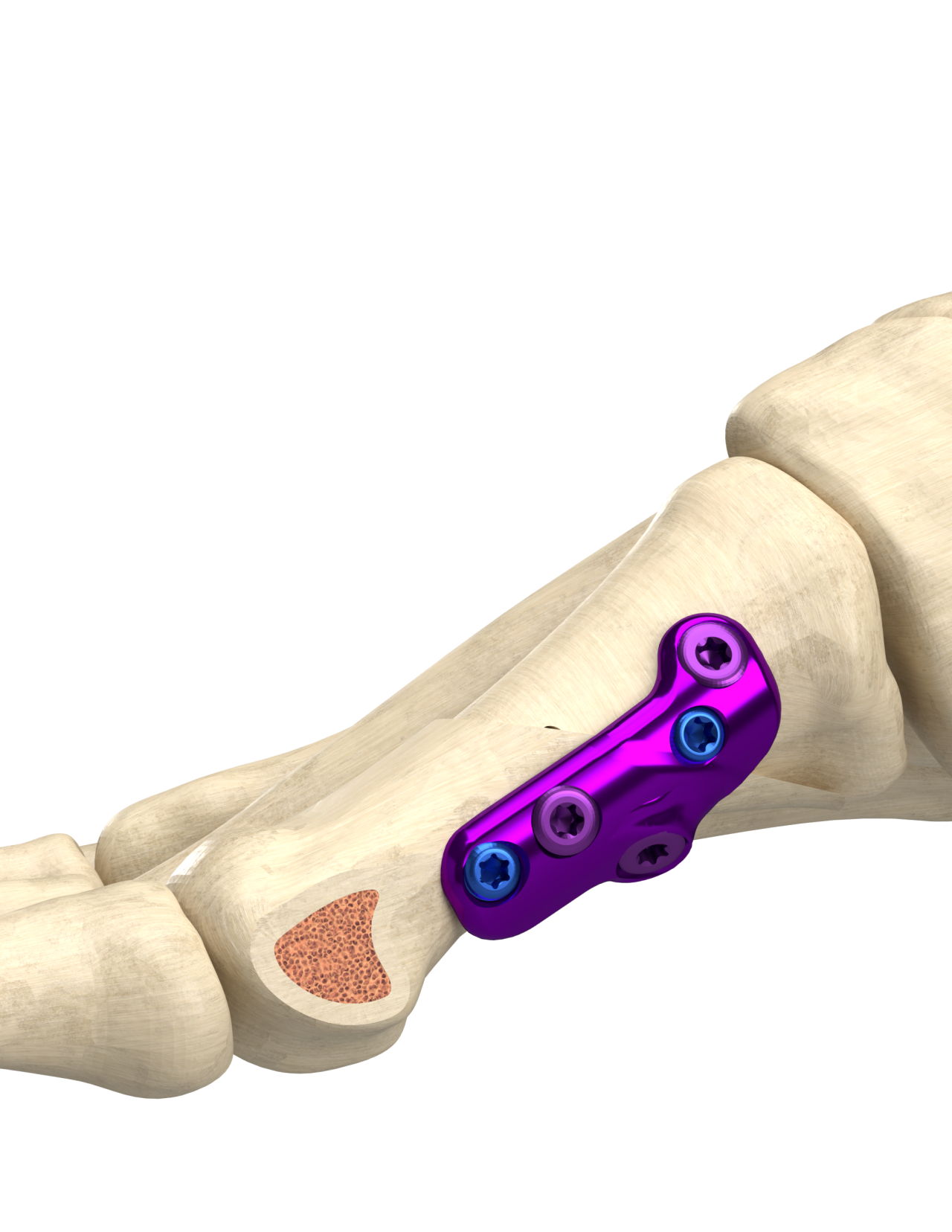
The Proximal Modified Mau Osteotomy:
- Provides superior intrinsic stability compared to most other midshaft, proximal osteotomies2-4
- Maximizes fusion surface area and provides the ability to correct moderate to large angular deformities2,5
- Avoids violating a healthy metatarsal-cuneiform joint compared to a Lapidus procedure
- Minimizes shortening of the metatarsal by eliminating the need to remove healthy bone while eliminating the need for interpositional grafts.5,6
Procedure Hallux Valgus (or Bunion)
Precise, Reproducible Osteotomy
Long Oblique Proximal Modified Mau Osteotomy
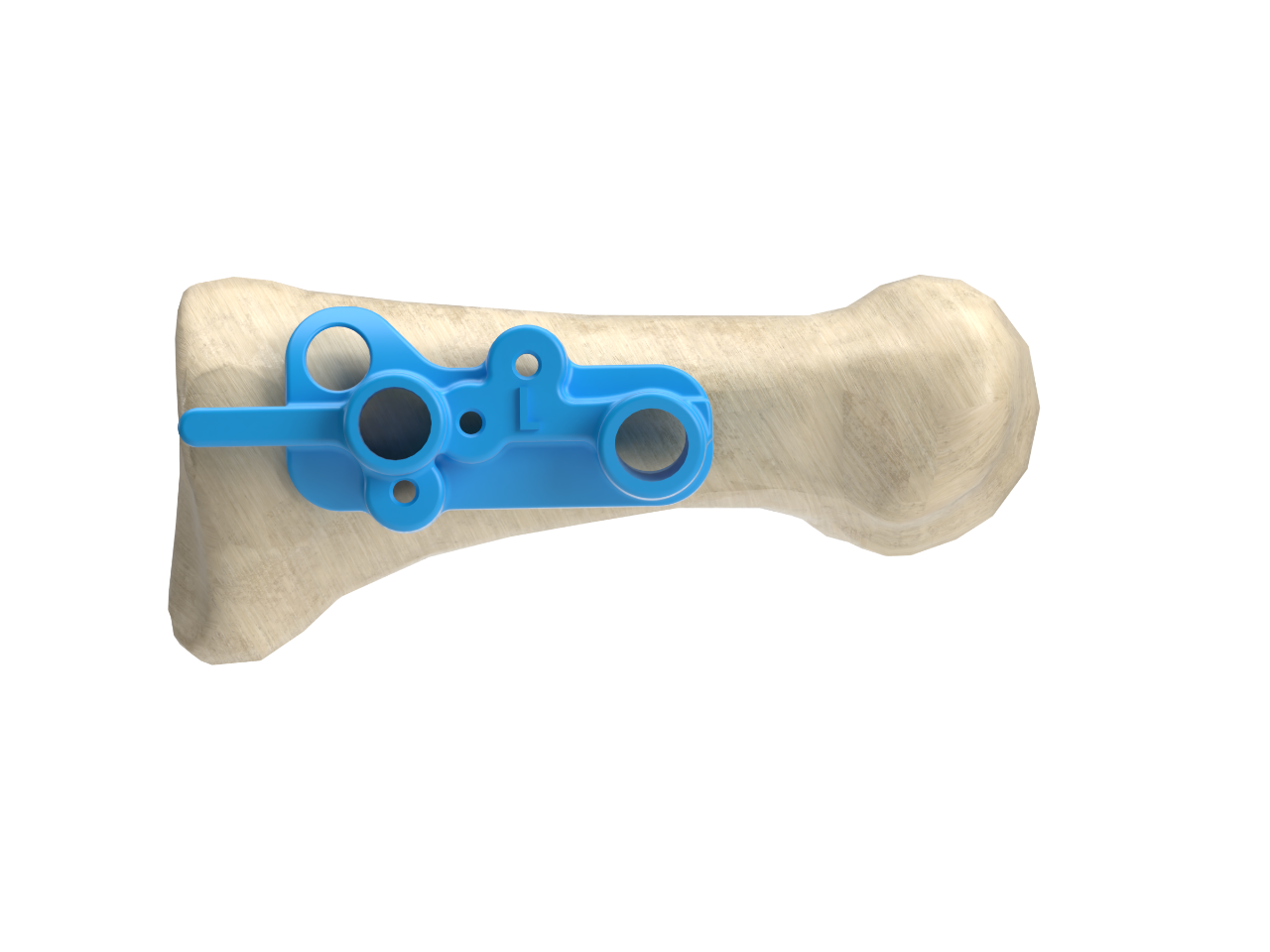
The system includes an Osteotomy Guide that provides a visible path for surgeons to cut a long oblique Proximal Modified Mau osteotomy.
The Axi+Line Proximal Bunion Correction System instrumentation provides full control of all bone fragments during the procedure and allows for a seamless transition from templating to plating.
System Features
The Axi+Line Proximal Bunion Correction System can be used to correct IM angle deformities of up to 18 degrees.
4 Left plates and 4 right plates
- Correction of 5°, 7.5°, 10°, 12.5°
Instrumentation
- Instrumentation is designed to provide full control of all bone fragments during the procedure and allows for a seamless transition from templating to plating.
All components in kits are pre-sterilized
- Instrumentation has never been used, damaged, or worn prior to surgery
- Time and cost savings as sterilization before surgery is no longer necessary
- Complete sets at delivery minimizes time at the back table
Videos
Axi+Line Proximal Bunion System - Surgical Animation
Watch animation
Frontal Plane Rotation
Watch animation
Additional Information
Tailored resources for your patients.
Find videos, articles, and interactive content to guide your patients throughout their surgical journey on ReadyPatient.com, our dedicated patient recovery site.
Legal Disclaimer
All content herein is protected by copyright, trademarks and other intellectual property rights, as applicable, owned by or licensed to Zimmer Biomet or its affiliates unless otherwise indicated, and must not be redistributed, duplicated or disclosed, in whole or in part, without the express written consent of Zimmer Biomet.
This material is intended for health care professionals. Distribution to any other recipient is prohibited.
For product information, including indications, contraindications, warnings, precautions, potential adverse effects and patient counseling information, see the instructions for use or contact your local representative; search this website for additional product information. To obtain a copy of the current Instructions for Use (IFU) for full prescribing and risk information, please visit labeling.zimmerbiomet.com or call 1-800-348-2759, press 4 for 411 Technical Support.