Medical Professionals
As a global medical technology leader, we push the boundaries of innovation to redefine what's possible across all stages of the patient journey.
Featured Products
Areas by Specialty
We seamlessly transform the patient experience through our innovative products and suite of integrated digital and robotic technologies that leverage data, data analytics and artificial intelligence.
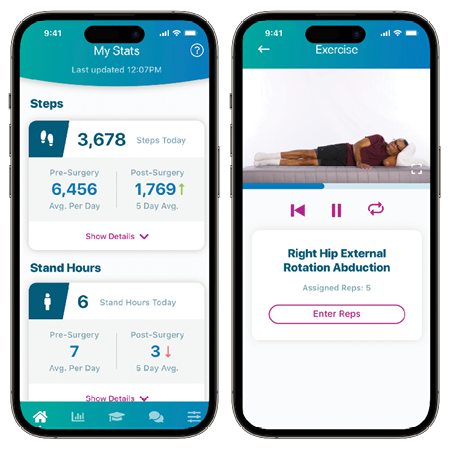
Explore New Possibilities with World-Class Education
We provide an interactive learning environment for enhanced education and training,while delivering personlized experiences.


Legal Disclaimer
All content herein is protected by copyright, trademarks and other intellectual property rights, as applicable, owned by or licensed to Zimmer Biomet or its affiliates unless otherwise indicated, and must not be redistributed, duplicated or disclosed, in whole or in part, without the express written consent of Zimmer Biomet.
This material is intended for health care professionals. Distribution to any other recipient is prohibited.
For product information, including indications, contraindications, warnings, precautions, potential adverse effects and patient counseling information, see the package insert or contact your local representative; search this website for additional product information. To obtain a copy of the current Instructions for Use (IFU) for full prescribing and risk information, please call 1-800-348-2759, press 4 for 411 Technical Support.
Canary Medical Logo, Canary Medical, CANARY canturio™te, and CHIRP™ are the exclusive property of Canary Medical.
For full prescribing information for the CANARY canturio™te, refer to physician instructions for use.
Patients must have compatible internet access and a text-capable mobile device, or a smartphone to use mymobility; not all smartphone app features are available with web-based version.
Not all patients are candidates for the use of this product and surgeons should evaluate individually to determine which patients are appropriate for therapy at home.
Legal Manufacturer: Canary Medical USA LLC, 2710 Loker Ave. West, Suite 350, Carlsbad, California 92010, USA, (833) 692-2627, www.canarymedical.com
Exclusive Distributor: Zimmer, Inc., 1800 West Center St., Warsaw, Indiana 46581-0587, USA, zimmerbiomet.com
All names used in the mymobility app examples are fictitious. No identification with actual patients or health care professionals is intended or should be inferred. Apple, iPhone and Mac are registered trademarks of Apple, Inc.