All-Suture Technology
JuggerKnot® Soft Anchor System – 1.0 mm Mini
Clinical Results, Versatile, and Strong
The introduction of the 1.4 mm JuggerKnot All-Suture Anchor in 2009 revolutionized soft tissue fixation in Sports Medicine, and we continue to innovate. This award winning1 100% suture based anchor system has quickly evolved into a family of anchors that offers one of the broadest ranges of options on the market for a multitude of different procedures.
Clinical Results
- One study of 18 patients, performed with the 1.4 mm anchor, demonstrated clinically improved patient outcomes and fibrous tissue, complete bony healing or combined fibro-osseous healing in the anchor tunnel2
Versatile
- Gain access to a robust soft anchor system on the market, which includes seven different size offerings and size specific instrumentation.
Strong
- High pullout strength ranging from 20-120 lbs., depending on size of anchor3,4
System Features
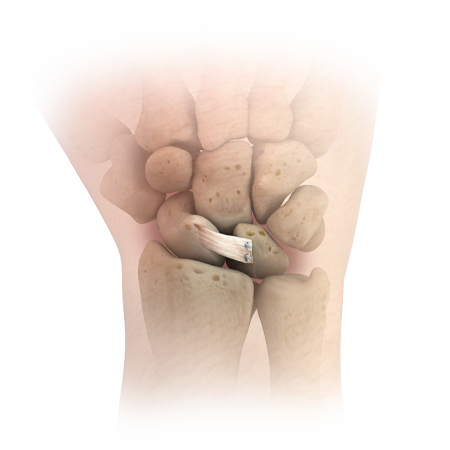
JuggerKnot Soft Anchor-1.0 mm Mini
-
Most commonly utilized for soft tissue to bone fixation in the hand
-
Designed with a #1 polyester sleeve, loaded with either a #2-0 or #3-0 MaxBraid™ suture, and loaded with two T-28 tapered needles
-
Sleeve protector eliminates the need for a drill guide
-
Currently the smallest all-suture anchor on the market
-
26 lbs. anchor pullout strength with #2-0 and 20 lbs anchor pullout strength with #3-0 suture4

Additional Information
Tailored resources for your patients.
Find videos, articles, and interactive content to guide your patients throughout their surgical journey on ReadyPatient.com, our dedicated patient recovery site.
All content herein is protected by copyright, trademarks and other intellectual property rights, as applicable, owned by or licensed to Zimmer Biomet or its affiliates unless otherwise indicated, and must not be redistributed, duplicated or disclosed, in whole or in part, without the express written consent of Zimmer Biomet.
This material is intended for health care professionals. Distribution to any other recipient is prohibited.
For product information, including indications, contraindications, warnings, precautions, potential adverse effects and patient counseling information, see the package insert or contact your local representative; search this website for additional product information. To obtain a copy of the current Instructions for Use (IFU) for full prescribing and risk information, please visit labeling.zimmerbiomet.com or call 1-800-348-2759, press 4 for 411 Technical Support.