Zimmer Biomet is a pioneer in Sports Medicine - our innovations have
directly helped move our industry forward and continue to do so
today. Our concise portfolio gives surgeons a comprehensive but
manageable solution set that can perform most any technique.
Plus, it's the only sports medicine solution set that's
integrative with our leading-edge technologies and continuum of care
in joint preservation, helping surgeons optimize caregiving for their
patients throughout the medical journey.
Sports Medicine Products
Pioneering. Integrative. Concise.
See our portfolio overview.
Pioneering Products
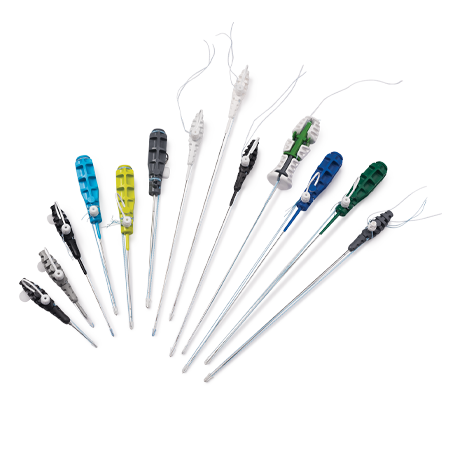
From the legendary JuggerKnot® All Suture Anchors to our all-in-one arthroscopy system, we continue to move our industry forward with innovation and excellence.
Rotator Cuff Repair
With a versatile rotator cuff portfolio, including knotless anchors, all-suture anchors, biocomposite anchors, traditional suture anchors, interpositional scaffold-anchor implants and suture passers, Zimmer Biomet has a variety of solutions for small to larger cuff tears. The following offering of anchors and accompanying passers allows surgeons to repair the rotator cuff, via double row or single row, knotless or with knots tied, to accommodate the tear pattern.
Shoulder Labral Repair
Labral tears vary from location to severity amongst patients so there is not just one solution. Find out more about our comprehensive implant and instrumentation offering for labral repair to accommodate each surgeon’s specific philosophy and technique providing versatility, strength, and clinical results.
Biceps Repair
The Zimmer Biomet portfolio provides a variety of options for mini-open or arthroscopic biceps repair procedures. Additionally, with suture anchors, suspensory fixation and tenodesis screws options for both on-lay and in-lay, there are a myriad of options available to make the best choice for the patient.
AC Joint Repair
Separated shoulder or AC joint injuries vary in severity amongst patients from acute to chronic in nature. Zimmer Biomet Sports Medicine provides a knotless, two-button suspensory fixation for clavicle reduction for arthroscopic or mini-open approaches.
Hand & Wrist
Zimmer Biomet offers the smallest anchor in the market for soft tissue reattachment in the hand and wrist with the JuggerKnot® 1.0 mm Mini Soft Anchor.
ACL Reconstruction
In knee arthroscopy, there are multitudes of ways to repair knee ligaments from graft choice to tunnel placement. Learn more below about the all-encompassing portfolio of implants, which provides solutions for both soft tissue and bone tendon bone grafts and all types of repairs including cortical and aperture fixation. Amongst the extensive offering of solutions are the first-to-market adjustable cortical fixation devices and PEEK interference screw.
Knee Instrumentation
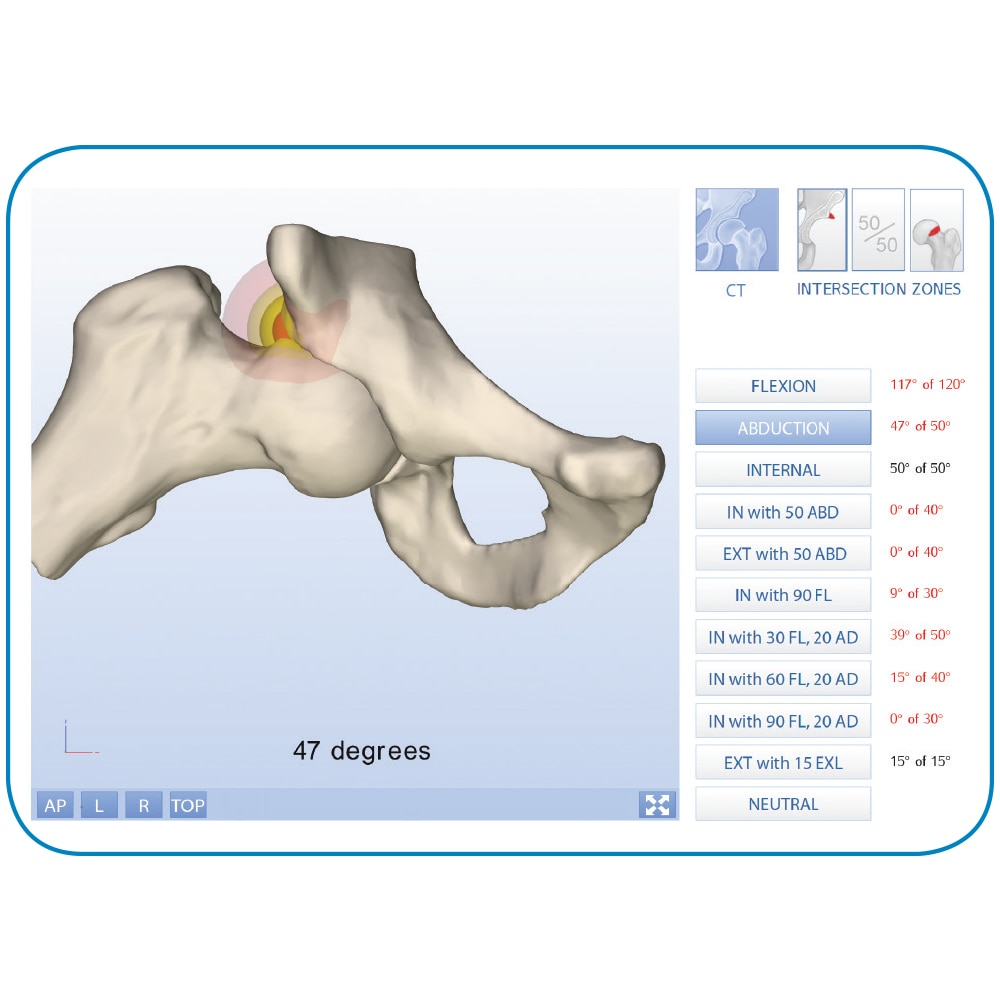
We are proud to offer an array of instrumentation for the numerous techniques to create socket and tunnels for multiligament reconstruction. These include the Anatomy Specific Guide, SwitchCut™ Retro-Reaming System, and Precision Flexible Reaming System. In conjunction with these great instruments, we also worked with surgeons who created the I.D.E.A.L.™1 ACL philosophy, which encourages the appropriate placement of the femoral tunnel.
- Howell, S., Pearle, A., & McAllister, D. (2015). Rationale for Strategic Graft Placement in Anterior Cruciate Ligament Reconstruction: I.D.E.A.L. Femoral Tunnel Position. American Journal of Orthopedics, 44(6), 253-258.
Meniscus Repair
Zimmer Biomet is proud to offer the JuggerStitch Meniscal Repair Device, an all-suture, all-inside, knotless meniscal implant.
Hip
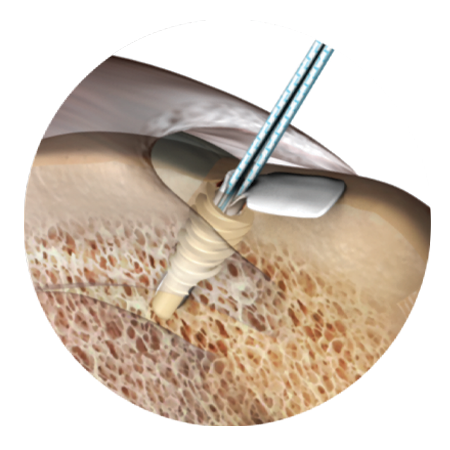
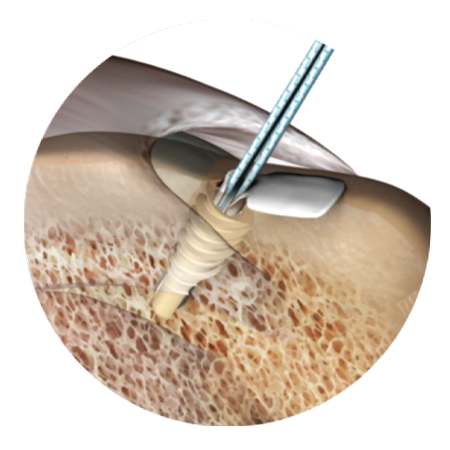
A comprehensive offering for hip acetabular labral repair incorporating the first-to-market and leading all-suture anchor system designed to prioritize strength and versatility in reaching difficult angles on the acetabular rim.
Arthroscopy & Imaging
Zimmer Biomet is excited to combine our comprehensive sports medicine portfolio with an innovative arthroscopy system option from RELIGN. Featuring an all-in-one device that combines fluid management, ablation, tissue resection, and bone cutting into a single versatile, proprietary bipolar shaver handpiece.
This combination fills an important gap in Zimmer Biomet’s sports medicine offering, allowing us to be a partner for all of your arthroscopic needs within the OR and providing the capability to completely equip an ASC.
Integrative Solutions
In Person and Online Training & Education
With our comprehensive medical education portfolio we aim to provide the knowledge you need to further pursue your goals of maximizing patient outcomes.
Tailored resources for your patients
Find videos, articles, and interactive content to guide your patients throughout their surgical journey on ReadyPatient.com, our dedicated patient recovery site.

All content herein is protected by copyright, trademarks and other intellectual property rights, as applicable, owned by or licensed to Zimmer Biomet or its affiliates unless otherwise indicated, and must not be redistributed, duplicated or disclosed, in whole or in part, without the express written consent of Zimmer Biomet.
This material is intended for health care professionals. Distribution to any other recipient is prohibited.
For product information, including indications, contraindications, warnings, precautions, potential adverse effects and patient counseling information, see the package insert or contact your local representative; search this website for additional product information. To obtain a copy of the current Instructions for Use (IFU) for full prescribing and risk information, please visit labeling.zimmerbiomet.com or call 1-800-348-2759, press 4 for 411 Technical Support.